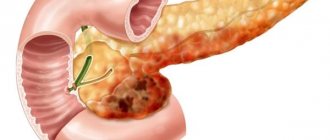
Acute myeloid leukemia (the most commonly used abbreviation is AML) is a malignant pathology of the blood and bone marrow, characterized by the uncontrolled production of immature (blast) myelocytic cells - red blood cells, granulocytes, neutrophilic leukocytes, platelets. The disease is diagnosed in cases where more than 20% of immature cells are detected in the myelogram. Its characteristic feature is its rapid course: within a few months, immature cells begin to predominate in the composition of the blood, which is why it ceases to perform its main functions. The patient's condition deteriorates sharply, and in the absence of treatment, the prognosis is extremely unfavorable.
Kinds
Oncohematologists distinguish several dozen types of acute myeloid leukemia, which are grouped according to similar characteristics:
- with typical genetic mutations;
- with dysplasia that causes cell changes;
- arising as a result of treatment of other diseases;
- with the proliferation of myeloid germ against the background of Down syndrome;
- myeloid sarcomas;
- dendritic cell plasmacytoid tumors.
Depending on the form of the disease, the doctor chooses treatment tactics, since the duration of remissions and the overall prognosis for different types have serious differences.
How are acute leukemias in adults classified?
Most tumors are classified into stages of disease (I, II, III, and IV), which are based on the size of the tumor and its extent.
This staging is not appropriate for leukemia because leukemia is a disease of the blood cells that does not usually cause tumor formation.
Leukemia affects the entire bone marrow and in many cases, by the time of diagnosis, has already involved other organs in the process. In leukemia, laboratory studies of tumor cells make it possible to clarify their characteristics, which help in assessing the outcome (prognosis) of the disease and choosing treatment tactics.
Three subtypes of acute lymphoblastic leukemia and eight subtypes of acute myeloid leukemia have been identified.
There are four main types of leukemia:
acute versus chronic
lymphoblastic versus myeloid
"Acute" means rapidly developing. Although the cells grow quickly, they are not able to mature properly.
"Chronic" means a condition where the cells appear mature but are actually abnormal (altered). These cells live too long and replace some types of white blood cells.
"Lymphoblastic" and "myeloid" refer to the two different types of cells from which leukemia arises. Lymphoblastic leukemia develops from bone marrow lymphocytes, myeloid leukemia arises from granulocytes or monocytes.
Leukemia can occur in both children and adults, but different types of leukemia predominate in one group or the other.
Acute lymphoblastic leukemia (ALL)
— Occurs in children and adults
- More often diagnosed in children
— Accounts for slightly more than half of all cases of leukemia in children
Acute myeloid leukemia (AML) (often called acute nonlymphoblastic leukemia)
- Affects children and adults
— Accounts for less than half of all cases of leukemia in children
Chronic lymphocytic leukemia (CLL)
- Occurs only in adults
— Detected twice as often as chronic myeloid leukemia (CML)
Chronic myeloid leukemia (CML)
- Mainly affects adults and is very rarely detected in children
— CLL is diagnosed twice as rarely.
Symptoms
In the initial stage of acute myeloid leukemia, there are practically no symptoms; as the disease progresses, the patient develops:
- pale skin and other signs of anemia;
- bleeding from the nose and gums, numerous bruises on the skin;
- elevated temperature within 37-38 degrees (subfebrile);
- profuse sweating at night;
- rash on the skin in the form of small reddish spots;
- shortness of breath even after light exertion;
- constant pain in bones and joints;
- frequent colds and other infections;
- enlarged lymph nodes;
- sudden weight loss for no apparent reason.
4.How to cure leukemia?
Acute myeloid leukemia
is a collection of oncological diseases, therefore the treatment of this disease depends primarily on the organ in which the accumulation of pathologically altered cells occurred, as well as on many other factors. Timely treatment is of great importance in oncology, so it should be started immediately after diagnosis.
In most cases, treatment for acute myeloid leukemia is a combination of methods, including radiation and chemical therapy, as well as bone marrow transplantation.
Chemotherapy
is based on the use of antitumor drugs: Prednisone, Cytarabine and other medications.
As a rule, this method underlies the treatment of cancer. Radiation therapy
uses high-energy radiation to kill cancer cells.
The peculiarity of this method is that only pathologically altered cells are exposed. A bone marrow transplant
involves transplanting bone-forming stem cells from a suitable donor.
Let's take a closer look at the treatment of acute myeloid leukemia. It is conventionally divided into several stages: induction of remission and consolidation. The goal of induction therapy is to achieve remission by destroying as many harmful cells as possible. Consolidation therapy is a therapy for maintaining remission. It is necessary to destroy the residual amount of abnormal blast cells in order to avoid relapse - recurrence of the disease.
What are the survival rates for acute myeloid leukemia? Over the past five years, this figure has increased by 24%. Such a positive prognosis indicates that this deadly disease can be defeated! The most important thing is to start treatment for acute myeloid leukemia on time and be regularly examined to prevent the recurrence of pathologically altered cells.
Causes and risk factors
It is currently unknown what exactly influences induce blood germs to malignant mutations, but oncologists have well studied the factors that contribute to the development of acute myeloid leukemia.
- Exposure to carcinogens. Certain chemical compounds can induce cells to change. These are a number of products formed during tobacco smoking and incomplete oxidation of fats, many types of industrial emissions, some medications, etc.
- Radiation. X-rays and other radioactive, ionizing radiation change the hereditary apparatus of living cells.
- Genetic factor. The risk of the disease increases several times for people who have close relatives with leukemia.
- Diseases. Some diseases increase the likelihood of blood cancer. These are Down syndrome, congenital anemia and thrombocytopenia, neurofibromatosis, etc.
Treatment of chronic myeloid leukemia
At the initial stage, before obtaining cytogenetic confirmation of the diagnosis (as we already know, chronic myeloid leukemia is diagnosed in the presence of the Philadelphia chromosome), symptomatic therapy with hydroxyurea is prescribed. Its goal is to reduce the overall level of white blood cells and platelets. If there is intolerance to the drug or insufficient reduction in platelet levels, anagrelide can be used. If there are signs of leukostasis (encephalopathy, visual disturbances, renal dysfunction), leukapheresis is performed.
After cytogenetic confirmation of the diagnosis, specific antitumor therapy is prescribed. The main drugs are tyrosine kinase inhibitors (TKIs). The dosage is selected depending on the level of leukocytes. At the initial stage, to prevent tumor lysis syndrome, enhanced hydration is necessary (additional fluid administration in a volume of 2-2.5 l/m2, if there are no contraindications from the cardiovascular system) and the administration of allopurinol.
The goal of specific treatment for chronic myeloid leukemia is to suppress the tumor cell clone, reduce the risk of pathology progression and prolong the patient’s life to values comparable to general population indicators. With the introduction of TKIs into practice, these tasks became quite feasible and not only made it possible to increase the overall survival of such patients several times, but also to eliminate lifelong medication use and transfer to dynamic observation in patients with a good molecular tumor response.
Currently, according to accepted treatment protocols, TKIs should be prescribed to all patients with newly diagnosed chronic myeloid leukemia. Their mechanism of action is based on the blockade of the ATP-binding pocket of the pathological molecule BCR-ABL, which deprives this protein of tyrosine kinase activity, which stimulates excessive division of tumor cells. With constant use of TKIs, the tumor clone undergoes reduction, which makes it possible to restore normal hematopoiesis.
In Russia, the following drugs from the TKI group are registered for first-line therapy:
- imatinib,
- nilotinib,
- dasatinib.
Imatinib
Imatinib has selective activity against BCR-ABL tyrosine kinase and several other tyrosine kinases. It is prescribed in long courses and must be taken daily. The initial dosage is 400 mg per day for the chronic phase of myeloid leukemia, and 600 mg/day for the accelerated phase or blast crisis. The dosage does not depend on the patient’s height, gender and body weight. The drug is available in tablet form or capsules. Can be used on an outpatient basis. If the result of therapy is unsatisfactory, the dosage can be increased, and if toxic complications develop, it can be reduced.
Nilotinib
Nilotinib is a highly selective BCR-ABL tyrosine kinase inhibitor. It was developed based on the imatinib molecule and modified to increase affinity for BCR-ABL tyrosine kinase. Available in capsules. The dosage for chronic phase therapy is 600 mg/day, for acceleration phase therapy - 800 mg/day. The drug is taken 2 times a day with an interval of 12 hours strictly on an empty stomach, since food increases the bioavailability of the drug, which increases its concentration in plasma and can provoke the development of toxic complications. If the therapeutic effect in the treatment of the chronic phase is insufficient, the dosage may be increased to 800 mg/day. If complications develop, the dose is reduced.
Dasatinib
Dasatinib has activity against many tyrosine kinases, including mutant BCR-ABL. Penetrates the blood-brain barrier. Available in forms for oral use. The recommended dosage is 100 mg/day for the chronic phase and 140 mg/day for the accelerated phase and blast crisis. If chronic phase therapy is insufficiently effective, the dosage may be increased to 140 mg/day. If toxic complications develop, it is reduced to 80 mg/day.
Bosutinib
Bosutinib is a relatively new drug, registered in Russia in 2014 and is used for second and subsequent line therapy in cases of intolerance or ineffectiveness of the above drugs. The standard daily dose is 500 mg, which can be increased to 600 mg if necessary.
The choice of first-line drug is carried out individually for each patient. This takes into account the phase of leukemia, the sensitivity of the tumor clone with individual mutations, the toxicity profile of each drug and the presence of concomitant diseases in the patient.
Determination of the spectrum of mutations is carried out when the pathology manifests itself in the acceleration phase or blast crisis, or when therapy with the selected drug is ineffective and there is a need to change the drug, since there is a possibility of the emergence of resistant clones. For example, mutations F317L/V, T315A, V299L cause low sensitivity to dasatinib, so it is changed to nilotinib. Mutations Y253H, E255K/V, F359V/C, on the contrary, make tumor cells resistant to nilotinib, so dasatinib is indicated for such patients.
Mutations E255K/V, G250E, V299L cause resistance to bosutinib. The presence of the T3151 mutation determines resistance to all types of TKIs, so allogeneic hematopoietic stem cell transplantation is recommended for such patients. Ponatinib therapy is also a possible option, but it has not yet been registered in Russia.
The effect of first-line therapy can be classified into one of three groups:
- Optimal answer. This is a good result, which allows us to hope for a long period of disease-free survival (7-8 years or more). The criteria for achieving optimal response are complete hematologic response within 3 months, complete cytogenetic response within 6 months, and major molecular response within 12-18 months.
- Warning. In the presence of warning factors, careful monitoring of the patient's condition and readiness to change the treatment regimen are required. Prevention factors include a high-risk group for chronic myeloid leukemia, a more than 10-fold increase in the transcription level of the mutant gene, and the presence of additional chromosomal abnormalities in cells with the Philadelphia chromosome.
- Therapy failure. This includes the progression of the disease, the emergence of new mutations, and the appearance of additional chromosomal abnormalities in cells with the Philadelphia chromosome. A change in therapy is required.
Other treatments for chronic myeloid leukemia
Donor stem cell transplantation is indicated for patients who have failed second-line therapy and for patients with the T315I mutation. Patients intolerant of TKIs and unable to undergo transplantation can be treated with hydroxyurea, interferons and cytostatics.
Diagnostics
Laboratory tests of blood and bone marrow samples are the main ways to diagnose acute myeloid leukemia. This:
- general peripheral blood test;
- myelogram;
- cytochemical analysis of immature cells;
- immunophenotyping;
- cytogenetic analysis;
- molecular genetic analysis;
- HLA typing;
- blood chemistry;
- Reberg's test;
- coagulogram.
In addition, the patient is prescribed instrumental examinations of internal organs to assess their condition: ultrasound of the abdominal cavity, ECG, chest x-ray. Subsequently, other tests and studies may be prescribed.
Diagnosis of chronic myeloid leukemia
In most cases, chronic myeloid leukemia is an incidental finding that is discovered during examination for another reason. It can be suspected by a general blood test, in particular by an increase in the number of leukocytes and the predominance of granulocytic hematopoiesis in the formula. In this case, not only the number of neutrophils, but also basophils and eosinophils may increase. There may be mild anemia or abnormal platelet counts.
If the doctor suspects chronic myeloid leukemia, the patient is referred for further examination - puncture and bone marrow biopsy. To confirm the diagnosis, it is necessary to conduct a standard cytogenetic study of the bone marrow for the presence of the Philadelphia chromosome. At least 20 metaphases are studied. If cytogenetics is not possible, fluorescent in situ hybridization is used to identify the chimeric gene. The expression of the chimeric gene in peripheral blood cells is also determined by PCR. If a typical transcript is not detected, and there are clinical and hematological signs of chronic myeloid leukemia, detection of rarer mutations is indicated - BCR-ABLp190, p230.
In the blast crisis phase, immunophenotyping of blast cells and cytological and biochemical examination of the cerebrospinal fluid are performed. When chronic myeloid leukemia is detected in the activation phase or blast crisis, the search for the Philadelphia chromosome can be carried out by sequencing the genetic material of blood cells.
Treatment
The main treatment method for acute myeloid leukemia is chemotherapy, which is carried out in two stages:
- induction - reducing the number of immature cells to achieve remission;
- consolidation - maximum prolongation of remission, prevention of relapses, elimination of residual effects.
Intensive therapy used during the induction phase allows achieving remission in approximately 70% of clinical cases. But treatment should absolutely not end there, since without a consolidation program, patients almost always experience a relapse after some time.
The consolidation stage, depending on the patient’s condition, includes up to five courses of chemotherapy. If the risk of relapse is high enough, a bone marrow and/or hematopoietic stem cell transplant is performed. The best donors are siblings, especially identical twins.
In addition, patients are often prescribed the administration of red blood cells and transfusion therapy (transfusions of blood and its drugs). Treatment tactics are usually chosen according to the patient's age. Remission is considered achieved if the number of myeloid cells in the bone marrow can be reduced to 20% or less.
charitable foundation
The essence of the disease
Leukemia, or leukemia, is a disease of the bone marrow, sometimes colloquially called “blood cancer.” In leukemia, normal hematopoiesis is disrupted: an excessive number of abnormal immature blood cells, usually the precursors of white blood cells, are produced. These blast cells, multiplying and accumulating in the bone marrow, interfere with the production and functioning of normal blood cells, which causes the main symptoms of the disease.
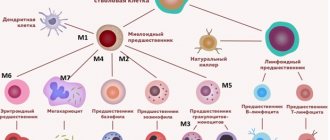
As you know, different blood cells develop differently and have different precursors - that is, they belong to different lines of hematopoiesis (see diagram in the article “Hematopoiesis”). The line of hematopoiesis leading to the appearance of lymphocytes is called lymphoid; the remaining leukocytes and other blood cells belong to the myeloid lineage. Accordingly, leukemias are distinguished from the precursor cells of lymphocytes (such leukemias are called lymphoblastic, lymphocytic, or simply lymphocytic leukemias) and from the precursors of other cells (such leukemias are called myeloblastic, myeloid, or simply myeloid leukemias).
Acute myeloid leukemia (AML, acute myeloid leukemia, acute myeloid leukemia, acute non-lymphoblastic leukemia) is a disease that is relatively rare in children, but its frequency increases with age. The term "acute" refers to the rapid progression of the disease, as opposed to chronic leukemia. The term “myeloid” (see above) means that the immature cells that form the basis of the disease belong to the so-called myeloid lineage of hematopoiesis. These cells are usually myeloblasts and their descendants, but the presence of other types of blast cells is also possible.
Within the framework of the French-American-British morphological classification (FAB), 8 main variants of AML are distinguished.
* M0 (AML with minimal differentiation): <5% of all AML cases * M1 (AML without maturation): 15-20% * M2 (AML with signs of maturation): 20-30% * M3 (Acute promyelocytic leukemia): 10- 15% * M4 (Acute myelomonocytic leukemia): 20-25% * M5 (Acute monocytic leukemia): 5-10% * M6 (Acute erythroid leukemia): <5% * M7 (Acute megakaryocytic leukemia): 3-10%
Some very rare variants of AML are not included on this list. Variants of AML from M3 to M7, which have their own names and some specific features, are discussed in more detail in separate sections of our reference book.
Frequency of occurrence, risk factors
AML accounts for about 15% of all cases of cancer of the hematopoietic system in children, that is, it occurs much less frequently in them than acute lymphoblastic leukemia. Among children under 14 years of age, the incidence of AML is approximately 0.6-0.8 cases per 100 thousand people per year, but after the age of 40-45 years there is a sharp rise in incidence. Most patients with AML are elderly. Unlike acute lymphoblastic leukemia, where the peak incidence is observed in childhood, with AML children make up only 10% of patients.
In most cases of AML, it is impossible to name the direct cause of the disease. However, some factors increase the likelihood of developing AML: exposure to a number of chemicals, ionizing radiation (including during previous treatment of other cancers). Sometimes cases of AML are observed among close relatives, which indicates a certain role of genetic predisposition. But specific genetic features that increase the risk of developing leukemia cannot yet be identified in many cases, although research is constantly being conducted.
The development of AML may be preceded by certain diseases of the hematopoietic system, such as myelodysplastic syndrome. Then they talk about secondary leukemia. Finally, the risk of AML is increased with certain genetically determined anomalies, including Down syndrome, Fanconi anemia, and others.
Signs and symptoms
AML is characterized by many different features and may present differently in different patients. The leading signs, as a rule, are anemia: fatigue, pallor, shortness of breath, loss of appetite. A lack of platelets is manifested by increased bleeding from cuts and bruises, nosebleeds, and the “unreasonable” appearance of bruises and hemorrhages. Treatment-resistant infections often occur because the patient has too few “normal” (mature) white blood cells to fight them. Damage to the mucous membranes of the mouth and gastrointestinal tract and swelling of the gums may occur. Body temperature is often elevated and bone pain is felt. Sometimes tumors arise from leukemic cells outside the bone marrow - myelosarcoma.
Since most symptoms can be associated with other diseases and are not specific to AML, before starting treatment it is necessary to clarify the diagnosis based on laboratory methods, which is urgently carried out in a hospital setting.
Diagnostics
With AML, changes occur in the usual clinical blood test: a lack of red blood cells and platelets, often an excess of white blood cells, many of them represented by immature forms. But a reliable diagnosis can only be made by examining a bone marrow sample. According to the World Health Organization criteria, the diagnosis of AML is made if the myeloblast content in the bone marrow is at least 20% (according to the French-American-British FAB classification, the threshold value is 30%).
To treat and assess the prognosis of the disease, it is important not only to confirm the diagnosis of leukemia, but also to distinguish between acute lymphoblastic leukemia and AML, to distinguish between leukemia and myelodysplastic syndrome, and also to determine the specific variant of AML (see above). For this purpose, not only morphological examination of cells is used (microscopic examination of specially stained preparations), but also cytochemical analysis, as well as immunophenotyping (study of antigen proteins on the surface of cells). Cytogenetic studies are used to detect chromosomal rearrangements - primarily translocations, which are of great importance for determining the variant of the disease and prognosis. Molecular genetic analyzes are also playing an increasing role.
Accurate diagnosis of AML and its variants is sometimes challenging, requiring the participation of highly qualified hematologists and hemopathologists in the diagnostic process.
As with acute lymphoblastic leukemia, the results of diagnostic tests for AML are used to assign a patient to a particular risk group. This is necessary in order to determine treatment tactics and assess both the likelihood of response to chemotherapy and the risk of subsequent relapse. Patients in the low-risk group are more likely to achieve long-term remission with standard chemotherapy treatment, while those at high risk will be considered for bone marrow transplantation to ensure the success of therapy.
The risk group is determined by many factors. Let's list some of them:
- Age: Middle-aged and elderly patients have a worse prognosis on average than children and young adults.
- Chromosomal changes in leukemic cells. Thus, translocations t(15;17) or t(8;21) determine a lower risk in patients. At the same time, for example, certain cytogenetic changes are associated with a worse prognosis.
- Variant of leukemia. Some variants of AML (such as M0, M6, M7) are associated with a high risk, and some, on the contrary, respond relatively well to modern therapy (AML M3).
- A higher risk is associated with secondary leukemia that occurs in the context of another hematological disorder, such as myelodysplastic syndrome or Fanconi anemia, or after treatment (chemotherapy, radiation) for a malignant tumor. The risk also increases when leukemia relapses.
Treatment
The main treatment for AML is chemotherapy. As in the case of acute lymphoblastic leukemia, treatment includes remission induction and consolidation phases; Sometimes maintenance therapy is also used.
Remission induction is intensive therapy aimed at achieving remission of leukemia. In most types of AML, induction of remission is carried out using intensive chemotherapy using cytarabine (Cytosar) and anthracycline drugs (daunorubicin, idarubicin), sometimes with the addition of other drugs - for example, etoposide or mitoxantrone.
A specific feature of therapy for acute promyelocytic leukemia (AML M3) is the use of the drug ATRA (all-trans retinoic acid) or other drugs with a similar effect (arsenic trioxide).
If, as a result of induction therapy, it turns out that there are less than 5% of blast cells in the bone marrow (against the background of restoration of hematopoiesis) and the patient has no other manifestations of the disease, then remission is stated.
As a result of induction courses, remission can be achieved in most patients. However, the achieved remission cannot be stable without consolidation therapy, that is, consolidation of remission. In the consolidation phase, residual amounts of abnormal blast cells are destroyed to avoid relapse of the disease. The most important drug in the consolidation phase of the treatment of AML is cytarabine, including high-dose; other drugs are often added to it in different combinations.
Chemotherapy regimens that include fludarabine are often used in the treatment of relapsed AML.
During the induction and consolidation stages, intravenous administration of chemotherapy drugs is performed in a hospital setting.
Maintenance therapy is not used in all cases (unlike acute lymphoblastic leukemia), but in the M3 AML variant it is important. This therapy is less intensive than induction and consolidation therapy and does not require a hospital stay.
Sometimes modern targeted drugs are added to therapy for AML. Specific medications depend on the genetic makeup of the tumor cells. For example, for a certain genetic disorder, the drug sorafenib is effective.
Neuroleukemia is less common in AML than in acute lymphoblastic leukemia. But sometimes it occurs, more often in children with AML variants M3, M4 and M5. For its treatment and prevention, cytarabine, methotrexate and glucocorticosteroids administered intrathecally through lumbar puncture of the spinal canal can be used.
To reduce the likelihood of relapse in some patients, bone marrow transplantation is indicated. It is used more often in AML than in acute lymphoblastic leukemia. Indications for transplantation may include, for example,
- a disease variant associated with a higher risk (see above),
- cytogenetic changes in leukemic cells associated with high risk,
- relapse of leukemia,
- secondary AML, which developed against the background of myelodysplastic syndrome or other blood disease, or after previous treatment of malignant tumors.
The likelihood of transplant success is greatest if it is performed after the first remission has been achieved. Transplantations are performed more frequently in children than in adults and are, on average, more successful. Work is constantly underway to improve transplantation regimens.
During intensive chemotherapy for AML, normal hematopoiesis is almost always suppressed to some extent. Therefore, many AML patients require transfusions of blood components: platelets to prevent bleeding and red blood cells to treat anemia.
Since both leukemia itself and the chemotherapy used in its treatment sharply reduce the body’s resistance to various infections, patients during treatment often need effective antibacterial, antifungal and antiviral drugs for the prevention and treatment of infectious complications. Both common and opportunistic infections pose a danger.
Treatment of AML imposes restrictions on the patient's lifestyle. During intensive chemotherapy, it is necessary to follow a diet and strict hygiene rules, as well as minimize contact with the outside world to avoid infections. Doctors and nurses tell each patient what he can and cannot do at the current stage of treatment.
The total duration of treatment for various forms of AML ranges from several months to (in the case of variant M3) 2-3 years.
New targeted drugs are constantly emerging to help cure patients with complex cases of AML. Thus, some patients benefit from venetoclax (Venclexta) and/or daratumumab (Darzalex). The conjugate drug gemtuzumab ozogamicin (Mylotarg) is sometimes used. In addition, bone marrow transplantation protocols are being improved.
Forecast
Without treatment, AML usually leads to death of the patient within several months, sometimes even several weeks. However, with modern treatment many can be saved. The prognosis depends on the specific type of leukemia, chromosomal characteristics of leukemic cells, age, general condition of the patient and other factors.
Currently, medicine in developed countries is able to cure more than 65% of children with AML. In particular, the results of bone marrow transplantation are constantly improving. In middle and old age, unfortunately, the likelihood of cure is significantly lower.
It should be noted that the majority of patients with AML are elderly patients. Compared to young people, they tolerate treatment more difficultly and respond less well to it. Only a minority of patients over 60 years of age achieve long-term remission. For others, supportive treatment (fighting infections, transfusions of blood components, pain relief) is often the mainstay to prolong life and improve its quality.