Avastin is an innovative antitumor drug that has recently appeared in the pharmacy assortment. The drug is in demand in oncology practice due to its ability to fight the most aggressive forms of cancer. Avastin contains recombinant humanized antibodies that are not toxic to the human body. But during treatment there is a possibility of developing adverse reactions. Therefore, it is advisable to use the drug only as prescribed by a doctor in the dosage regimen specified by him.
Characteristics of Avastin
Avastin is a product of genetic engineering. Its composition is formed from nucleic acids, which were obtained by combining foreign fragments with nucleotides. This combination is considered in medicine and pharmacology to have properties as close as possible to the cells of the human body. Monoclonal antibodies selectively combine with the bioactive factor VEGF, which leads to its rapid neutralization. The malignant tumor gradually decreases in size and then disappears altogether.
Avastin is available in the form of a fine crystalline powder. This is a concentrate that is diluted with an isotonic sodium chloride solution before use. The powder is packaged in glass bottles with rubber stoppers and aluminum caps of 4 or 16 ml. The cardboard box also contains an annotation, the recommendations of which will help to carry out the treatment correctly.
Material and methods
Description of samples
5 samples of Avastin were received. All samples were clearly labeled indicating the name of the drug, active substance, concentration, dosage form, production date, batch, expiration date; All bottles had the company logo on the label. The bottles did not have protective aluminum caps or plastic lids and were sealed with rubber stoppers. The samples received for examination were from the same production series. The original Avastin drug of the same series, seized from the manufacturer, was used as a reference sample for the comparative study.
The samples received for examination were assigned internal laboratory numbers from No. 1 to No. 5, the reference sample was No. 6.
Physico-chemical characteristics
The relative density of the contents of bottles with the drug "Avastin" was determined using an Atago PAL-10S refractometer (Japan), and the pH of solutions was determined using a universal pH meter Seven Compact S220, Mettler-Toledo AG (USA).
A preliminary test for the presence of protein in the preparations was carried out by adding an organic solvent (cold acetone) to the test solution in a solution-acetone ratio of 1:5. The formation of a precipitate was assessed as a positive result. Deionized water was used as a negative control, and reference sample No. 6 was used as a positive control.
Protein concentration was determined by UV-SFM using a NanoDrop ND-1000 UV spectrophotometer at 260/280 nm and a molar extinction coefficient of 243.340 M–1cm–1 for 1 mg/ml. The drug filtrate obtained after filtration on 10 kDa Amicon Ultra 10K centrifuge filters (Millipore, USA) was used as a negative control (blank).
Common reactions to authenticity
For qualitative analysis of anions, we used filtrates obtained after centrifugation of the contents of the studied preparations on 10 kDa Amicon Ultra 10K filters.
The presence of chloride ions in the samples was determined by adding dilute nitric acid and silver nitrate (GF XII) to the test sample. To confirm the presence of chloride ions, a standard sodium chloride solution was used, and deionized water was used as a negative control.
The presence of phosphate ions in the samples was determined by reaction with silver nitrate (SP XIII, method A) or with ammonium molybdate (SP XIII, method B). To determine the quantitative content, phosphates were converted to phosphorus-molybdenum blue, which absorbs radiation in the visible region of the spectrum. To do this, the samples under study were diluted 1000 times with deionized water, after which the resulting solution was filtered through 3 kDa Macrosep Advance 3K centrifugal filters (Pall Corporation, USA) to remove protein. Then, 10 μL of sulfomolybdenum reagent was added to 500 μL of the filtrates of each sample. After 5 min, 5 μl of a freshly prepared tin(II) chloride solution was added to the samples and left for an additional 10 min to form molybdenum blue. The resulting colored solutions were subjected to spectrophotometric (SPM) analysis (Cary 60 UV-Vis spectrophotometer, USA) at a radiation wavelength of 690 nm in a cuvette with an optical layer length of 10 mm. To prepare calibration standards, samples of sodium hydrogen phosphate were taken containing known concentrations of phosphate ion in the range of 0.75-10 μg/ml. Calibration curves were obtained by analyzing two series of calibrators, each of which contained six calibration solutions. Each series of calibrators was analyzed twice. To describe the parameters of the calibration functions, a linear model was used, constructed by the least squares method (LS) in the coordinates: X is the concentration of the analyte under study in the calibration standard (μg/ml), Y is the optical density of the analyte solution. The Pearson correlation coefficient (R2) for the phosphate ion calibration function was 0.9997.
Mapping of bevacizumab peptides by high-performance liquid chromatography-high-resolution mass spectrometry (HPLC-MS/HRMS)
Enzymatic hydrolysis protocol
Samples
were analyzed by HPLC–MS/HRMS after proteolytic digestion with trypsin . An aliquot of the test sample (40 μl) was diluted with a solution of 8 M urea (GE Healthcare, Sweden) in 50 mM Tris-HCl (Sigma-Aldrich, USA) to a theoretical bevacizumab content of 1 mg/ml (960 μl). 10 μL of the resulting solutions were transferred into 0.5 ml tubes (Eppendorf LoBind, Germany), and an additional 15 μL of 8 M urea in 50 mM Tris-HCl (pH 7.4) was added. To restore disulfide bonds, 2.5 μl of a 100 mM solution of 1,4-dithiothreitol (DTT, Sigma-Aldrich, USA) was added to each sample, thoroughly mixed and incubated at 56 °C for 1 hour with constant stirring on a thermomixer. (Eppendorf, Germany). After this, the reduced disulfides were alkylated by adding 2.5 μl of 200 mM iodoacetamide (Sigma-Aldrich, USA) to each sample; the reaction mixture was thoroughly mixed and kept in the dark at room temperature for 45 min. Then, 2 μL of 100 mM DTT solution was re-added to the samples and kept in the dark for an additional 10 min at room temperature. After reduction and alkylation of disulfides, the volume of each sample was adjusted to 100 μl with 50 mM ammonium bicarbonate (pH 7.8), then 200 ng/μl of trypsin working solution (Sequencing Grade Modified Trypsin; Promega, USA) was added in a trypsin-to-protein mass ratio 1:20. The reaction mixture was thoroughly mixed by vortex and then incubated overnight at 37 ºC. Termination of the trypsinolysis reaction was carried out by adding concentrated formic acid to each sample to a final concentration of 5%. Samples were transferred into vials and analyzed by HPLC–MS/HRMS.
HPLC-MS/HRMS parameters
The analysis of tryptic hydrolysates of the studied samples of the drug "Avastin" was carried out by HPLC-MS/HRMS using a hybrid liquid chromatography-mass spectrometer LCMS-IT-TOF, equipped with pumps model LC-20AD, degasser model DGU-20A5R, autosampler model SIL-20ACXR, column thermostat model CTO-20AC, as well as a control module CBM-20A combined with an IT-TOF mass spectrometer (Shimadzu Corp, Japan). Separation of tryptic peptides was carried out on an analytical column Phenomenex Kinetex C18 column (150×2.0 mm; 2.6 μM) using gradient elution while thermostatting the analytical column (37 °C) in the following mode: 0 min - 5% B, 5 min - 10% B, 15 min - 40% B, 17 min - 98% B, 19 min - 98% B, 19.5 min - 5% B, 25 min - 5% B. As aqueous (A) and organic (B) The mobile phases used were 0.1% formic acid in water and 0.1% formic acid in acetonitrile, respectively. The total analysis time was 25 min, the flow rate of the mobile phases was 0.4 ml/min. Analytes were ionized in an electrospray ionization source under the following conditions: nebulizer gas flow rate (N2) 1.5 L/min, desolvation line temperature 200 °C, heating block temperature 250 °C, interface and detector voltage 4.5 and 1.6 kV respectively. MS/MS spectra were obtained by collision-induced dissociation with an inert gas (argon) in automatic mode (auto ms2). The precursor ion was selected in the range of 300–1500 m/z, the precursor ion isolation window was 3 m/z. The ion accumulation time was 20 and 50 ms for the MS1 and MS/MS stages, respectively, the relative collision energy was 50%, and the volume of analyzed samples was 3 μL.
The data obtained were collected and processed using LC/MS Solution software, version 3.41. Identification of proteotypic peptides was carried out by processing HR mass spectra using PEAKS Studio software, version 7.0.
Determination of electrophoretic mobility, assessment of the content and purity of bevacizumab by CAP-PAGE under reducing conditions
CAP-PAGE analysis followed by staining with Coomassie G-250 solution (Cat # 161−0803, Bio-Rad, USA) was performed to determine the amount of the drug in the studied samples No. 1-5 compared to its content in the reference sample No. 6, as well as to assess the position of bevacizumab protein bands in the gel under denaturing conditions before and after IdeS hydrolysis and determine their molecular weights (mol. wt). For the experiment, 10 μg of each test sample of the drug and the reference sample (No. 1-6) were taken in 10 μl of phosphate-buffered saline (PBS). For hydrolysis in the presence of IdeS, 20 U of protease (stock solution in water, 25 U/μL) was added to each sample and incubated in a thermomixer at 37 °C for 1 h (350 rpm; Eppendorf, Germany). Then add 3.5 µl of 4x sample loading buffer (424 mM Tris hydrochloride, 564 mM Tris base, 8% sarkosyl, 40% glycerol, 2.04 mM EDTA and phenol red as lead dye), 0.5 µl of 40-fold reducing agent 1,4-dithiothreitol and heated in a thermomixer at 95 °C for 5 min (350 rpm; Eppendorf, Germany). MOPS buffer solution (50 mM MOPS, 50 mM Tris base, 0.1% sarkosyl, 1 mM EDTA) was used as an electrode buffer solution. Next, all samples were added to the wells of a polyacrylamide pre-filled 12% Bis-Tris Precast gel in Criterion format (Cat # 345−0118, Bio-Rad, USA) along with samples of pre-stained standards mol. m. (7 µl, Cat # 161−0393, Bio-Rad, USA). Electrophoresis was carried out at a constant voltage (180 V) for 60 min using an electrophoretic cell of the Criterion format (Cat # 165−6001; Bio-Rad, USA). The gel was then washed for 3 × 5 min with deionized water and incubated in Coomassie G-250 solution for 45 min. Next, the gel was washed for 5×10 min with deionized water and the colored protein bands were visualized using the ChemiDoc™ MP gel documentation system (Bio-Rad, USA).
What is Avastin for?
The main indication for treatment with Avastin is malignant neoplasms, including those complicated by metastases. Oncologists prescribe the drug to patients after diagnosing the following pathologies:
- colorectal cancer - a tumor that forms in the rectum, as part of complex therapy;
- inoperable forms of lung cancer in combination with drugs containing platinum compounds;
- malignant neoplasms affecting renal structures, together with immunomodulators alpha-2a interferons;
- focal cancer localized in the fallopian tube, ovary, one of the abdominal organs in combination with Carboplatin or Paclitaxel.
Avastin is also included in therapeutic regimens when other anticancer drugs are ineffective.
Cancer immunotherapy in Israel
Russian Medical Server / Cancer immunotherapy in Israel / Avastin (Bevacizumab) - cancer treatment in Israel
Innovative drug Avastin for immunotherapy in Israel
Avastin is a medicine that prevents the vascular system from supplying cancer tumors with nutrients and oxygen. Thanks to this process, it becomes possible to change the formation from malignant to chronic. In turn, this makes it possible to perform operations on tumors that previously could not be removed.
General information about the medicine
Avastin (Bevacizumab) is an antibody produced by immune cells. As mentioned above, the substance interferes with VEGF (vascular endothelial growth factor). This factor is the main one in triggering the growth of the number of vessels that feed the tumor.
In addition to stopping the expansion of the circulatory system, Avastin is also used to control tumors and their metastasis (recurrence of cancer).
Story
The development of drugs that prevent antiogenesis (supply of tumors through blood vessels) began quite recently, at the end of the 20th century. The first successful experience of therapy with such drugs was recorded in 1989. Then, since 1992, active research and development of similar drugs for the treatment of malignant tumors began. In 1997, the results of some experiments were published in the journal Nature; they showed a decrease in cancer formations after treatment with antiogenic drugs. A year after that, scientists created the first laser to prevent the formation of blood vessels for tumors, and a year later the first course of antiangiogenic therapy was developed. It was approved by the FDA, and after that even more active research and development began in this area. In 2003, Avastin (Bevacizumab), the first antiangiogenic drug, was created and showed good patient survival.
A number of studies have confirmed that the drug increases survival for any type of tumor. The drug is approved in Russia and Europe for the treatment of four main types of cancer in the final stages - cancer of the rectum and colon, breast, lungs and kidneys. According to statistics, about three million people die from these diseases every year.
In the USA, the medicine is approved by the FDA (Food and Drug Administration). In North America, the drug is used to treat five types of cancer - breast, colorectal, kidney and lung.
Currently, about 500 thousand people have received treatment with Avastin. The international company Roche, which is the largest drug manufacturer in the world, annually sponsors research on the drug. In total, more than 455 studies of the drug were conducted on seven types of malignant tumors at different stages of development.
Indications and contraindications for use
Unfortunately, Avastin cannot be used in all cases. The drug is prescribed for intestinal cancer, when the tumor has begun to spread to other organs (metastatic type of disease, characteristic of the last stage of the disease). In this case, the medicine is prescribed in combination with chemotherapy.
This remedy is also prescribed in the case of a malignant tumor of the mammary glands, when the cancer has developed into a locally recurrent form (re-formation of a tumor in the same place). In this case, treatment is carried out in combination with taxane-based chemotherapy. This method of treatment is used as first-line therapy.
Another indication for Avastin is relapse of lung cancer. The medicine is used if surgery cannot be performed. This therapy is also used primarily from the moment of diagnosis. The complex also includes chemotherapy based on platinum substances.
Another medicine is prescribed for renal cell carcinoma. With this disease, damage to the epithelium of the kidney occurs. In this case, Avastin is taken in combination with interferon Alpha 2a.
The drug is also prescribed for brain tumors (glioblastoma). This type of education is considered the most dangerous. These patients should take Avastin as their primary medication or in combination with irinotecan if the person relapses.
The medicine also has some contraindications.
The drug should not be taken:
· during pregnancy;
· during breastfeeding;
· if you are allergic to any components of the drug;
· if you are allergic to medications made using Chinese hamster ovary cells and other similar components;
· if the patient is a child (safety and effect are unknown);
· in case of kidney or liver failure (safety and effect unknown).
Avastin should also be taken with caution in case of arterial hypertension accompanying cancer, or clogging of blood vessels with blood clots. There are some restrictions for people over 65 years of age, so the use of the medicine in very elderly patients should be carefully monitored. It is not yet advisable to take Avastin during the healing of a serious wound or stopping bleeding. In addition, it is not recommended for patients with stomach or intestinal ulcers, as well as people with serious diseases of the heart and vascular system.
Side effects and overdose
Patients may experience some side effects from this drug. For example, a gastrointestinal ulcer, bleeding into the lungs, coughing up blood (most often occurs in people with a tumor in the lungs), the appearance of blood clots in the vessels. In addition, increased blood pressure, weakness, abdominal pain and diarrhea are possible.
The vascular system can also react to taking the medication. Many patients experience a decrease in white blood cells, neutrophils in the blood, and anemia.
As mentioned above, an ulcer may appear while taking the drug. In addition, from the gastrointestinal tract, nausea, stool disorders, anorexia, intestinal obstruction, etc. are possible.
It is also possible to change the shade of the skin - redness, dermatitis, etc.
As for the nervous system, a change in taste perception, migraine, increased drowsiness, etc. may occur.
In addition, increased lacrimation and visual impairment are noted as a side effect of the medication.
The maximum dose of Avastin is 20 mg/kg. If an overdose occurs, all of the above effects may be enhanced.
Features of application
Firstly, the drug is taken strictly under the supervision of a doctor. In addition, it should be remembered that Avastin slows down wound healing. So if an operation has been performed, then you need to start the course of treatment no earlier than a month after the surgery. If any complication occurs, it is better to stop taking Avastin.
The drug can be taken only after measuring blood pressure. If a person has hypertension, then while the condition stabilizes, you need to stop taking the medication for a while. If there is a risk of crisis, it is not recommended to take the medicine. If the patient has a history of hypertension, then the amount of protein in the urine may increase.
If a person has poor blood clotting or chronic hemorrhagic diathesis, the drug should also be taken with caution. You also need to carefully monitor doses if the patient has lung cancer or there is a risk of excessive hemoptysis. They can be caused by treatment with anti-inflammatory drugs, radiation therapy, the development of a tumor in the center of the lung, or the deposition of fatty plaques in the blood vessels. If a person regularly coughs up 2.5 ml. blood and higher, then Bevacizumab should be stopped.
If the cancer is in the intestines, there may be bleeding from the rectum while taking the medicine. In addition, during the studies, blood discharge from the nose was observed. Typically the nosebleed lasted about 5 minutes or less. This occurred in 40% of people. Increased gum bleeding is also possible.
Despite the fact that the drug had high survival rates, Avastin provokes blockage of blood vessels with blood clots (thromboembolism), a heart attack. Moreover, the risk of both occurrences is much higher than when prescribing chemotherapy alone. If the patient is over 65 years of age, thromboembolism occurs even more often, so doses must be prescribed very carefully.
Sometimes a person may develop late-onset encephalopathy (destruction of the white matter of the brain that affects mental abilities). In this case, you should stop taking Avastin completely and monitor your blood pressure. Unfortunately, doctors cannot say for sure what might happen if Bevacizumab treatment is reintroduced later.
If a patient is diagnosed with breast cancer, there is a high risk of congestive heart failure. So in the case of a history of cardiovascular disease, you need to think very seriously about the doses and consequences.
Quite often, after a course of Avastin, a person with an intestinal tumor developed fistulas (inflammation) in the stomach or intestines. In rare cases, a fistula may appear in other parts of the body. Most often, inflammation appears after six months of taking Bevacizumab, but sometimes doctors diagnose it after a year or even one week of therapy. If a fistula appears in the gastrointestinal tract, the person should stop taking the medicine. In case of inflammation in other parts of the body, cessation of therapy is decided individually.
If Avastin is taken in combination with other chemotherapy drugs that interfere with the process of hematopoiesis, neutropenia (a decrease in the number of neutrophils in the blood) or infection may develop. Sometimes a patient can be diagnosed with both at the same time, in such cases the risk of death is high.
When taking Avastin by people over 65 years of age, there is a high risk of arterial blockage with blood clots leading to the development of stroke, heart attack and micro-stroke. Diarrhea, headache, weakness, etc. are also possible.
General data based on Avastin studies
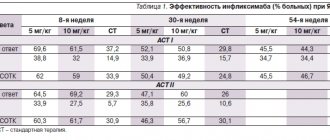
In the ECCO and ESMO studies, it was proven that the drug in combination with chemotherapy (based on Xeloda) reduces and completely eliminates liver metastases in 80% of patients suffering from metastatic (the tumor has spread to neighboring organs) colon cancer. Therefore, a third of patients with malignant tumors can undergo surgery to remove metastases. As a result, about 60% of people were successfully operated on.
The BOXER study was also conducted, which studied the body's response in patients with stage 2 liver metastases to Avastin in combination with XELOX (a drug based on Xeloda, taken orally, Oxaliplatin is administered intravenously in the complex). The result was also positive - patients who took Xeloda had a higher survival rate than those who underwent standard chemotherapy. After this, XELOX complex was also given to patients with stage three colon cancer. As a result, 71% of people achieved three-year survival; the group of patients taking a different course of chemotherapy showed the same survival rate in only 66.5% of cases.
The First BEAT study showed that the drug is equally effective in young and elderly patients. This is a very important discovery, because... The elderly population rarely appears in such experiments.
According to statistics, colon and rectal cancer is the third most common disease (about 600 thousand people die from this disease every year). In addition, this is the second most dangerous cancer disease (after brain cancer), so reducing the risk of relapse with the help of Avastin in combination with Xeloda will please many.
There is also a study NO16968, during which scientists observed the effects of the XELOX therapeutic complex for six months. About 2,000 people suffering from colon cancer took part in the experiment. NO16968 was conducted in 29 countries and 226 research centers. The main result of the study showed that people's survival rate increased without the risk of tumor recurrence.
How Avastin works
Its course use prevents the formation of additional blood vessels that feed the tumor. It stops increasing in size, growing, and metastasizing. The therapeutic effects of Avastin are due to the properties of its active ingredient bevacizumab, which represents a clinical and pharmacological group of drugs with recombinant monoclonal antibodies. Under its influence, the receptors of growth factors of the first and second types, which are located on the surface of the endothelium, are blocked. First, vascularization decreases, and after some time a decrease in the tumor is noted. How much does the anticancer drug Avastin cost? Go to the corresponding section of the site to find out.
Pharmacokinetics
The maximum therapeutic concentration of the active substance Avastin is ensured by its parenteral administration once every 1-2 times a month. The age of the patient does not affect the clearance of bevacizumab. It is almost a third higher in patients whose blood contains low levels of albumin. The active ingredient is metabolized by proteolytic catabolism by all cells of the body.
Instructions for use
You can find out the cost of the antitumor drug from our catalog. Avastin is used as an injection solution for intravenous administration. During the preparation of the medicine, compliance with the rules of asepsis is required. When calculating single doses, the oncologist takes into account the type of cancer, tumor size, and severity of symptoms. He focuses on the patient’s age and the presence of other chronic diseases. When the concentrate is mixed with an isotonic solution, a drug is obtained in which the content of bevacizumab varies between 1.4–16.5 mg/ml. There are several nuances to Avastin therapy:
- The first treatment procedure cannot last less than one and a half hours. If no adverse reactions occur, then the next intravenous administration lasts no more than an hour.
- The injection solution is administered not by junior medical personnel, but by an oncologist with practical experience in treating cancer with monoclonal antibodies.
- Throughout the treatment, the patient's condition is monitored based on functional indicators of the liver and kidneys.
No dosage adjustment is required for elderly and senile patients. But it is necessary for people with liver and kidney pathologies.
Avastin in ophthalmology
Avastin is a medical drug used in the treatment of ophthalmic diseases since 2004. It was developed as an oncological agent that can slow down the growth of tumors and reduce their volume for subsequent surgical treatment. However, in the course of clinical studies, another ability was noted - to slow down the growth of newly formed vessels, which is now successfully used in ophthalmology in the treatment of age-related macular degeneration and other eye diseases accompanied by abnormal proliferation of blood vessels (neovascularization).
Thus, injections of Avastin into the vitreous body for proliferative diabetic retinopathy turned out to be very effective and well tolerated by patients. In addition, it is highly effective in stabilizing the development of the subretinal neovascular membrane - the growth of newly formed vessels in the fundus.
Ophthalmological practice of using Avastin shows that in the majority of patients, after treatment there was a significant improvement in the functional state of the organ of vision. The drug is especially effective in cases of hemorrhages into the vitreous body, with neovascularization of the iris. Intravitreal administration of the drug is also used in the treatment of wet forms of macular degeneration in the elderly, macular edema, diabetic retinopathy, and neoplasms of the perimacular region.
The effect of Avastin administration is expressed in the thinning of the macular region of the retina and stabilization of the subretinal neovascular membrane, which is confirmed by fluorescein angiography, which shows a decrease in fluorescein extravasation.
Injections of the drug into the vitreous minimize the risk of developing systemic side effects due to microdosing, which is sufficient for a targeted effect (400-500 times less than the dose used for intravenous administration), however, even this allows the doctor to create the necessary concentration of the substance in a given area. Avastin in a dosage of 1.25 mg is injected into the vitreous body once every 3-4 weeks. The maximum effect is often observed immediately after the first injections.
Since the beginning of the use of Avastin, there have been positive changes in the treatment of the wet form of age-related macular degeneration. This drug has high effectiveness and affordable cost. The response to treatment with it, as studies show, does not depend on photodynamic therapy and the administration of the drug Macudzhen - the results will be the same as with treatment carried out for the first time. In addition, there are no age restrictions when treating AMD with Avastin.
The limitations of using the drug intraocularly include only childhood, pregnancy, and the period of breastfeeding, due to the rather painful procedure for intraocular administration.
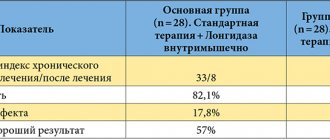
Over the past years since the drug was first used in ophthalmology, a large number of scientific papers have been published regarding Avastin therapy methods, with an analysis of the results obtained in the treatment of patients with the exudative form of AMD. The conclusion of these studies is clear: thanks to the administration of Avastin, there is a significant increase in visual acuity in almost a third of patients, and in more than half of patients, visual acuity is stabilized.
Contraindications to treatment with Avastin
An absolute contraindication to antitumor therapy is hypersensitivity to one of the components. Avastin is used with caution in the treatment of patients with the following pathological conditions:
- hypertension;
- thromboembolism;
- hemorrhagic diathesis;
- blood clotting disorders;
- severe cardiovascular diseases;
- agranulocytosis;
- history of perforation of ulcers;
- acquired coagulopathy;
- proteinuria.
In pediatric practice, treatment with Avastin is prohibited. This is also true during pregnancy and breastfeeding. For patients with severe renal or liver failure, safer drugs are selected.
Side effects of Avastin
Very rarely, the following serious adverse reactions were observed: gastrointestinal perforation, hemorrhage, including pulmonary hemorrhage/hemoptysis (more often in patients with non-small cell non-squamous cell lung cancer), arterial thromboembolism. The increase in blood pressure and the development of proteinuria is likely to be dose-dependent. In patients who received Avastin, hypertension (arterial hypertension), asthenia, diarrhea and abdominal pain are most often detected. The following are side effects of any severity observed in patients receiving Avastin or combination treatment with chemotherapy. From the cardiovascular system : hypertension (arterial hypertension), arterial thromboembolism (including myocardial infarction, stroke, transient ischemic attacks and other arterial embolisms); deep vein thrombosis, chronic heart failure, supraventricular tachycardia, bleeding. From the hematopoietic system : leukopenia, neutropenia, anemia, thrombocytopenia. From the gastrointestinal tract: abdominal pain, diarrhea, constipation, rectal bleeding, stomatitis, bleeding gums, gastrointestinal perforation, intestinal obstruction. From the respiratory system : nosebleeds, shortness of breath, rhinitis, hypoxia, pulmonary thromboembolism. . Skin: palmoplantar erythrodysthesia syndrome, dry skin, exfoliative dermatitis, skin discoloration. From the central nervous system : taste distortion, anorexia, syncope, headache, stroke, drowsiness, peripheral sensory neuropathy. From the organ of vision: visual impairment . Local reactions: pain at the injection site. Others : asthenia, abscess, sepsis, fever, vaginal bleeding, urinary tract infections, fatigue, pain of various localizations, dehydration, anorexia. Laboratory Abnormalities Grade 3 and 4 laboratory abnormalities observed in patients receiving Avastin with or without chemotherapy included proteinuria, hypokalemia, hyperkalemia, hyponatremia, hypophosphatemia, hyperglycemia, neutropenia, thrombocytopenia, increased prothrombin time, increased international normalized ratio . Side effects registered during the post-registration period Very rare: perforation of the nasal septum; hypertensive encephalopathy with fatal outcome. Rarely: reversible late leukoencephalopathy syndrome, including epileptic seizures, headache, mental disorders, visual impairment, damage to the visual center, measles, hypertension. Common: dysphonia.