Leakage of amniotic fluid
When choosing a management plan for a pregnant woman, the gestational age is first taken into account. Taking into account the non-viability of the fetus and the high risk of dangerous complications, it is recommended to terminate pregnancy before 22 weeks. At a later stage, patients are hospitalized in level 3 obstetric hospitals and are provided with maximum physical rest. From 22 to 34 weeks inclusive, the tactics are expectant. Prolongation of pregnancy is contraindicated in case of chorioamnionitis, gross malformations, acute hypoxia or fetal death, onset of labor, preeclampsia and eclampsia, placental abruption and a number of other conditions. In other cases it is recommended:
- Preventive antibacterial therapy
. To reduce the risk of infectious complications, antibiotics are prescribed in a course from the first day of hospitalization. - Drugs for the prevention of RDS
. To accelerate the synthesis of surfactant and the maturation of lung tissue, glucocorticoids are used. - Tocolytic therapy
. The use of selective β2-adrenergic agonists, magnesium sulfate, cyclooxygenase inhibitors, blockers of slow calcium channels and oxytocin receptors reduces the contractile activity of the myometrium. - Amniotransfusion
. It is used in exceptional cases at the request of a pregnant woman at 22-24 weeks with large volumes of discharge and a significant decrease in the amount of amniotic fluid.
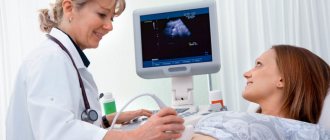
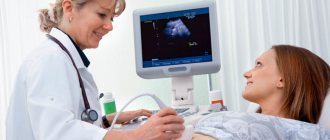
Treatment is carried out against the background of regular monitoring of the condition of the fetus and the pregnant woman: thermometry, assessment of blood parameters and hemodynamics, daily cardiotocography, weekly ultrasound, Doppler measurements of fetoplacental blood flow every 3 days. In this case, special attention is paid to the prevention of nosocomial infections.
When detecting water leakage in a woman with a 34-36 week pregnancy, both expectant and active tactics are used. Since there is no reliable evidence that induction of labor at this stage worsens neonatal outcome, the second option is preferable. Waiting often provokes the development of chorioamnionitis and compression of the umbilical cord. The duration of observation usually does not exceed 1 day. After the onset of labor, antibiotics are prescribed for prophylactic purposes.
At 37 or more weeks of gestation, if amniotic fluid is detected in the discharge and labor is absent, induction of labor is indicated. Antibacterial therapy is used in the diagnosis of chorioamnionitis. Preventive antibiotics are recommended only in cases where the expected duration of water leakage exceeds 18 hours.
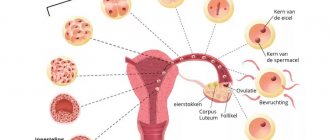
What are the functions of amniotic fluid during fetal development?
Throughout pregnancy, amniotic fluid provides reliable protection for the fetus from injury, bruises, blows and other mechanical damage. Besides
- this is a source of comfortable habitat for the embryo
- amniotic fluid is intensively involved in metabolism
- they protect the umbilical cord from compression
- serve as a barrier that prevents the fusion of the skin of the fetus with the walls of the uterus
- protect the child’s body from various infections from the genital tract
- help prepare the baby’s gastrointestinal tract, respiratory and excretory systems for work outside the mother’s body
CONDUCTING RESEARCH
- Take the test tube with the solvent by the cap and shake well, making sure that all the liquid inside the test tube sinks to the bottom. Open the tube and place it in a rack in a vertical position.
- To collect a sample from the vaginal surface, use the sterile polyester swab included in the kit. Remove the sterile swab from the package according to the instructions on the package. Before insertion into the vagina, the tip of the tampon should not come into contact with anything. Take the tampon by the middle of the handle and carefully insert the polyester tip of the tampon into the vagina to a depth of no more than 5 - 7 cm. Leave the tampon in the vagina for 1 minute, and only then remove it.
- Dip the polyester end of the swab into the tube and rinse the swab in the solution by rotating the swab for 1 minute.
- Remove the swab and place it in a disinfectant solution for treating biological waste.
- Open the foil pouch at the cut points and remove the AmniSure test strip.
- Dip the white end of the test strip (indicated by arrows) into the solvent tube and leave it for at least 5 and no more than 10 minutes . Massive leakage of amniotic fluid will make the result visible early (after 5 minutes), and small leakage will become visible only after 10 minutes.
- Remove the test strip from the tube if both stripes appear (but not earlier than 5 minutes) or exactly 10 minutes after immersion. Read the results by placing the test strip on a clean, dry, flat surface. Do not read the results if more than 15 minutes have passed since the test strip was immersed in the tube.
When to see a doctor
If you are pregnant full term and your water breaks, stay calm and contact your doctor or maternity hospital to find out when you need to leave. You should also consult a doctor in the following cases:
- The waters broke before the 37th week
of pregnancy
, - Contractions don't start
although the waters have already broken. In this case, your doctor may recommend inducing labor.
In case the doctor recommends that you go to the maternity hospital immediately, you should have a bag packed with all the necessary things.
Amniotic fluid index - upper and lower limits of normal
The volume of fluid in the amniotic sac is assessed by a medical specialist during all routine ultrasound scans. For each week of pregnancy, the IAF table contains data on the normal fluid volume along with maximum permissible deviations from these indicators. If the AFI value exceeds the upper limit of normal, the doctor can conclude that there is polyhydramnios. A slight excess of the norm means moderate polyhydramnios.
| Gestational age in weeks | Average normal value in mm | Probable fluctuations in mm |
| 16 | 121 | 73-201 |
| 17 | 127 | 77-211 |
| 18 | 133 | 80-220 |
| 19 | 137 | 83-225 |
| 20 | 141 | 86-230 |
| 21 | 143 | 88-233 |
| 22 | 145 | 89-235 |
| 23 | 146 | 90-237 |
| 24 | 147 | 90-238 |
| 25 | 147 | 89-240 |
| 26 | 147 | 89-242 |
| 27 | 156 | 85-245 |
| 28 | 146 | 86-249 |
| 29 | 145 | 84-254 |
| 30 | 145 | 82-258 |
| 31 | 144 | 79-263 |
| 32 | 144 | 77-269 |
| 33 | 143 | 74-274 |
| 34 | 142 | 72-274 |
| 35 | 140 | 70-279 |
| 36 | 138 | 68-279 |
| 37 | 135 | 66-275 |
| 38 | 132 | 65-269 |
| 39 | 127 | 64-255 |
| 40 | 123 | 63-240 |
| 41 | 116 | 63-216 |
| 42 | 110 | 63-192 |
Clinical trial results
Sensitivity: 99 % Specificity: 100% . Positive diagnostic value: 100% . Negative diagnostic yield: 99% .
In parallel with the nitrazine test and the crystallization test, patients were studied using AmniSure. If there were discrepancies between AmniSure and other tests, the patient was placed under close clinical observation. In all cases of such discrepancies, the AmniSure test results were found to be more accurate. The study protocol described above resulted in two sets of data: “adjusted results” and “unadjusted results.” Unadjusted results were collected on the assumption that the AmniSure test gave incorrect results when inconsistent with other tests. After carefully monitoring all of these cases and confirming that the AmniSure test was correct in all cases of discrepancy, a “corrected” data set was compiled. As a result, the AmniSure test was much more accurate than other tests studied.
ADVANTAGES OF THE TEST
The AmniSure kit is a self-contained test system that provides quality results that are superior in speed, accuracy, sensitivity, specificity and reliability to all existing methods. The AmniSure test covers the entire spectrum of diagnostic needs, from simple cases where a confirmatory test is simply needed, to the most complex cases where there is no visible leakage of amniotic fluid (so-called subclinical rupture). The test does not require insertion of a speculum, a practice now widely used to diagnose ruptured membranes.
PRECAUTIONS AND WARNINGS
- The AmniSure test kit is for in vitro diagnostic use only. No components should be ingested.
- Read and follow the instructions exactly. Failure to do so may result in inaccurate results.
- When collecting biomaterial, working with the test and working with waste, precautions must be observed.
- Damaged test components should not be used.
- The tests used are biohazardous.
- Take necessary precautions when handling the tests used.
- Do not use tests after the date stated on the kit packaging and box.
- Do not reuse test components.
- Do not bend or crease the test strip (dipstick) or the aluminum foil package that contains the test strip.
REAGENTS AND COMPONENTS
Set contents:
- Instructions.
- AmniSure test strip (lateral flow device, “dipstick”) in a foil pouch with desiccant. Each test strip contains: a) monoclonal antibodies A produced by mouse hybridoma; b) immobilized monoclonal antibodies B produced by another mouse hybridoma; c) colloidal gold particles associated with monoclonal antibodies A; d) labeled mouse IgG; e) Rabbit IgG against mouse antibodies.
- Sterile polyester vaginal tampon.
- Plastic test tube with aqueous solvent. The solution contains: a) 0.9% NaCl; b) 0.1% Triton X100; c) 0.01% NaN3.
Cross reactivity
The specificity of the monoclonal antibodies used in AmniSure was studied by examining their cross-reactivity with the following proteins: α2-fertility microglobulin, human chorionic gonadotropin, trophoblastic β1-glycoprotein, human placental lactogen, α-fetoprotein, human serum albumin and certain insulin-like growth factors. Monoclonal antibodies A and B used in AmniSure did not react with these proteins, with one exception: antibodies B cross-reacted with insulin-like growth factor BP-1 using the ELISA method. It has been shown that IGFBP-3 concentrations in the vagina of pregnant women can reach 680 ng/ml, but this did not affect the results of the AmniSure test.